Problem
A leading solid oxide fuel cell (SOFC) manufacturer required a low-cost catalyst that can operate in their portable, light-weight fuel cell for sustainable electricity generation.
Why is it a challenge?
Fuel cells convert chemical energy to electrical energy through a pair of redox reactions. Generally, hydrogen is used as the fuel and oxygen is used as the oxidiser. Solid oxide fuel cells can be operated on a variety of other fuels converted (with a dry reforming process) into hydrogen.
Direct internal reforming of the fuels provide low overall system costs, less weight and complexity due to the elimination of the external reformer. Fuel cells equipped with an integrated internal reforming catalyst layer will lead to simpler designs and higher fuel efficiency. To be suitable for this process, the catalyst must offer high conversion to hydrogen, in order to maintain the supply to the anode. The catalyst must also be thermally stable in cold-hot cycling and withstand high temperatures (above 700 °C) over prolonged periods of time (>100 hours).
What did we do?
Stoli tested a variety of catalyst formulations to determine an optimum catalyst that provided high yields of hydrogen. We used our proprietary coating methodology to obtain a uniform coating on a high temperature stable monolith support for low pressure drop allowing the unrestricted flow of catalytically-formed hydrogen to the fuel cell’s anode.
We also used radiofrequency heating of the catalyst to perform rapid cold-hot cycles to assess the catalyst stability. Using radiofrequency heating compared to conventional heating allowed rapid heating and cooling rates (well above 200 oC/min) for thorough worst-case scenario testing carried out in a fraction of conventional experiment time.
Outcome
Stoli developed stable catalyst samples for the fuel cell manufacturer to improve the overall fuel efficiency and pave the way for small-scale sustainable electricity generation. Application of radiofrequency heating allowed rapid testing of catalyst stability towards cold-hot cycles.
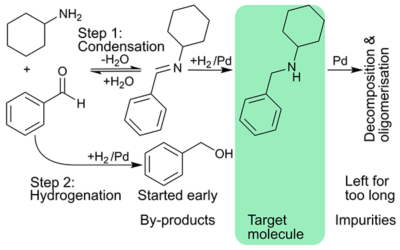
Cascading imine formation and hydrogenation
Stoli cascaded imine formation and hydrogenation; intensified process to maximise rate, and catalyst utilisation.
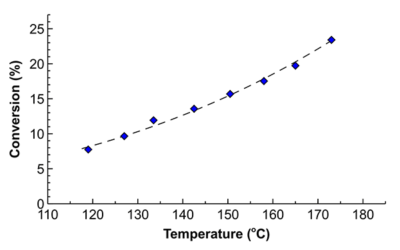
Process intensification in hydrogenation
Short residence time and high temperature – an impossible combination for batch – allowed increasing specific reaction rates 8-fold in flow with the same product quality.