Problem
A customer aimed to improve the activity and stability of an industrial catalyst establishing understanding relationships between the catalyst recipe, its structure, and performance.
Which analysis?
Stoli studied coordination and oxidation state of the active catalyst component in situ under reaction conditions. We selected X-ray absorption spectroscopy (XAS) as the method for determining the oxidation state, coordination chemistry, and coordination number of the studied element. High sensitivity and rapid data acquisition allowed detailed in situ study.
XAS studies can only be performed using synchrotron radiation due to the requirement of high-intensity X-ray radiation of a broad spectrum.
XAS is typically split into two categories:
(i) X-ray absorption near-edge spectroscopy (XANES) which is sensitive to the oxidation state and coordination chemistry
(ii) Extended X-ray absorption fine structure (EXAFS) which can determine the distances and coordination number of the neighbouring atoms.
What did we do?
At a synchrotron, every beam time minute is precious. Stoli team worked round the clock to perform 10 reduction and oxidation experiments in 24 hours to characterise the catalyst under the industrial conditions.
Outcome
The XAS analysis uncovered subtle changes in redox behaviour. It was possible to determine and monitor the change in oxidation state as temperature is increased. The data showed that the bulk material does not change its coordination or oxidation state, but the surface oxidation state changes.
Correlation of in situ oxidation with catalyst behaviour and recipe allowed the improving catalyst performance.

Experimental system used for synchrotron studies
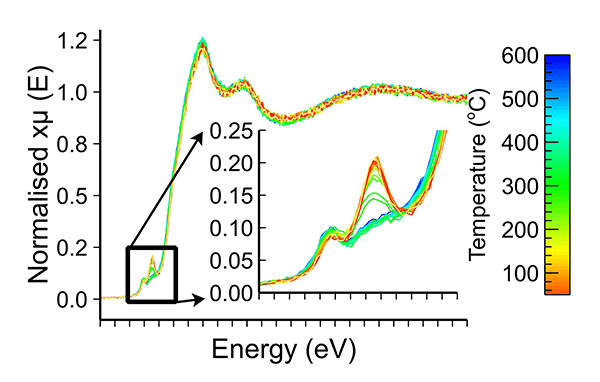
X-ray absorption spectra of a reference catalyst during in situ temperature-programmed oxidation
Catalyst for fuel cell applications
We have developed a monolith-based fuel cell catalyst for sustainable remote energy generation.
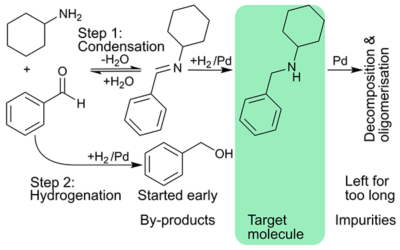
Cascading imine formation and hydrogenation
Stoli cascaded imine formation and hydrogenation; intensified process to maximise rate, and catalyst utilisation.
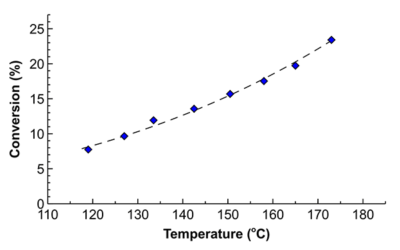
Process intensification in hydrogenation
Short residence time and high temperature – an impossible combination for batch – allowed increasing specific reaction rates 8-fold in flow with the same product quality.